|
1/8/2024 0 Comments Blood brain barrier definition
The ExAblate MRI-guided FUS (MRgFUS) system consists of a high-field MRI scanner, a hemispheric 1,024-element phased array ultrasound transducer interfaced with computer systems to align, steer, and control the transducer array utilizing skull data from a computed tomography (CT) scan ( Fig. The ExAblate Neuro system (InSightec Ltd.) and the NaviFUS system are both devices for use through the intact skull ( 11– 13). Modern FUS systems include devices that can activate circulating microbubbles (MBs) to create temporary BBBO through the intact skull. To date, there are no methods for effective, noninvasive, and safe BBB opening (BBBO) with the potential for spatiotemporal control of localized drug delivery in brain tumors ( 3, 6– 9).įocused ultrasound (FUS) is an emerging technology capable of noninvasively delivering acoustic energy into tissues throughout the body ( 10). This vexing challenge of limited drug delivery across the BBB in infiltrating gliomas motivated the work described here. Glioblastoma (GBM, grade IV glioma), a particularly aggressive and invasive form of brain cancer, is associated with a grim prognosis in part due to the lack of permeability of the BBB to otherwise promising therapeutics ( 4, 5). Thus, the BBB effectively limits pharmacologic treatment of numerous CNS diseases, including infiltrating gliomas, which diffusely invade the brain ( 3). 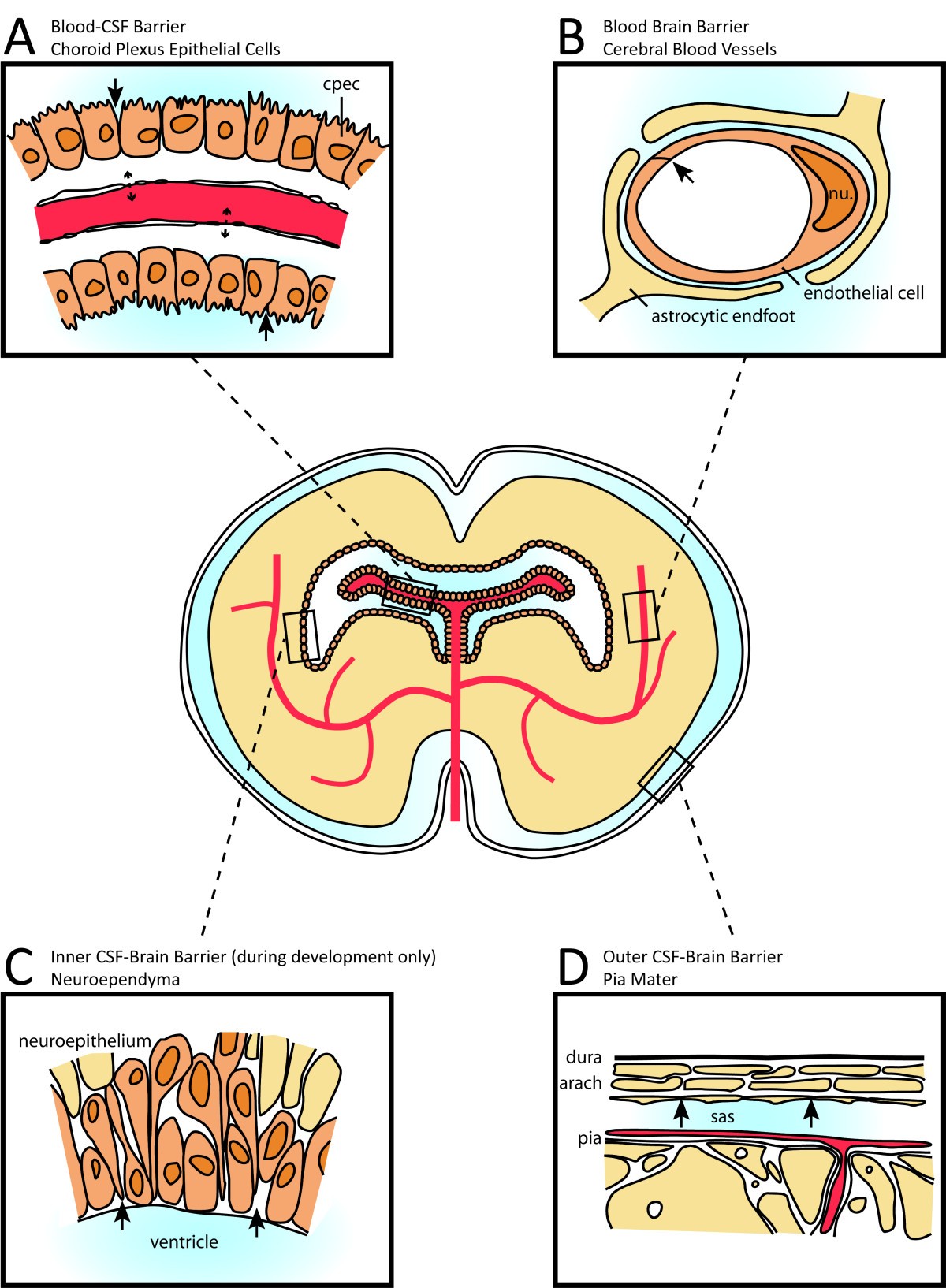
The blood–brain barrier (BBB) is composed of neurovascular units with contributions from endothelial cells, pericytes, astrocytes, and neurons that together control the trafficking of metabolites, signaling molecules, toxins, pathogens, and most drugs between the bloodstream and central nervous system (CNS) ( 1, 2). Collectively, this study demonstrates the capabilities of MB-FUS for safe, localized, controlled BBB opening and highlights the potential of this technology to improve the surgical and pharmacologic treatment of brain tumors. Importantly, we identified a 2.2-fold increase of fluorescein accumulation in MB-FUS–treated compared to untreated nonenhancing tumor tissues ( P < 0.01) while accounting for vascular density. We found a positive correlation between microbubble acoustic emissions (HD) and MR-evident BBB opening ( P = 0.07) and associated interstitial changes ( P < 0.01), demonstrating the unique capability to titrate the MB-FUS effects in gliomas. This range underscores the need for monitoring and titrating the exposure on a patient-by-patient basis. These observations occurred using a power cycling closed feedback loop controller, with the power varying by nearly an order of magnitude on average. Contrast-enhanced MRI and histopathology indicated safe and reproducible BBB opening in all patients. In this Phase 0 clinical trial (NCT03322813), we conducted comparative and quantitative analyses of FUS exposures (sonications) and their effects on gliomas using MRI, histopathology, microbubble acoustic emissions (harmonic dose ), and fluorescence-guided surgery metrics. Here, we evaluate the safety and utility of transcranial microbubble-enhanced FUS (MB-FUS) for spatially targeted BBB opening in patients with infiltrating gliomas. Transcranial MRI-guided focused ultrasound (FUS), an emerging technology for noninvasive brain treatments, enables transient opening of the BBB through acoustic activation of circulating microbubbles. Pharmacological treatment of gliomas and other brain-infiltrating tumors remains challenging due to limited delivery of most therapeutics across the blood–brain barrier (BBB).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
